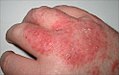
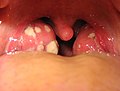
Inflammation

Inflammation (Latin, inflammare, to set on fire) is part of the complex biological response of vascular tissues to harmful stimuli, such as pathogens, damaged cells, or irritants.[1] Inflammation is a protective attempt by the organism to remove the injurious stimuli and to initiate the healing process. Inflammation is not a synonym for infection, even in cases where inflammation is caused by infection. Although infection is caused by a microorganism, inflammation is one of the responses of the organism to the pathogen.
Without inflammation, wounds and infections would never heal. Similarly, progressive destruction of the tissue would compromise the survival of the organism. However, chronic inflammation can also lead to a host of diseases, such as hay fever, atherosclerosis, rheumatoid arthritis, and even cancer (e.g., gallbladder carcinoma). It is for that reason that inflammation is normally closely regulated by the body.
Inflammation can be classified as either acute or chronic. Acute inflammation is the initial response of the body to harmful stimuli and is achieved by the increased movement of plasma and leukocytes (especially granulocytes ) from the blood into the injured tissues. A cascade of biochemical events propagates and matures the inflammatory response, involving the local vascular system, the immune system, and various cells within the injured tissue. Prolonged inflammation, known as chronic inflammation, leads to a progressive shift in the type of cells present at the site of inflammation and is characterized by simultaneous destruction and healing of the tissue from the inflammatory process.
Causes
- Burns
- Chemical irritants
- Frostbite
- Toxins
- Infection by pathogens
- Physical injury, blunt or penetrating
- Immune reactions due to hypersensitivity
- Ionizing radiation
- Foreign bodies, including splinters, dirt and debris
- Trauma
Types
| Acute | Chronic | |
|---|---|---|
| Causative agent | Pathogens, injured tissues | Persistent acute inflammation due to non-degradable pathogens, persistent foreign bodies, or autoimmune reactions |
| Major cells involved | Neutrophils, mononuclear cells (monocytes, macrophages) | Mononuclear cells (monocytes, macrophages, lymphocytes, plasma cells), fibroblasts |
| Primary mediators | Vasoactive amines, eicosanoids | IFN-γ and other cytokines, growth factors, reactive oxygen species, hydrolytic enzymes |
| Onset | Immediate | Delayed |
| Duration | Few days | Up to many months, or years |
| Outcomes | Resolution, abscess formation, chronic inflammation | Tissue destruction, fibrosis, necrosis |
Cardinal signs
| English | Latin | |
|---|---|---|
| Redness | Rubor* | |
| Swelling | Tumor* | |
| Heat | Calor* | |
| Pain | Dolor* | |
| Loss of function | Functio laesa** | |
| All the above signs may be observed in specific instances, but no single sign must, as a matter of course, be present.[2]
These are the original, or "cardinal signs" of inflammation.[2]* Functio laesa is an apocryphal notion, as it is not unique to inflammation and is a characteristic of many disease states.[3]** | ||

Acute inflammation is a short-term process, usually appearing within a few minutes or hours and ceasing upon the removal of the injurious stimulus.[4] It is characterized by five cardinal signs:[5]
The acronym that may be used for this is "PRISH" for Pain, Redness, Immobility (loss of function), Swelling and Heat.
The traditional names for signs of inflammation come from Latin:
The first four (classical signs) were described by Celsus (ca 30 BC–38 AD),[7] while loss of function was added later by Galen[8] even though the attribution is disputed and the origination of the fifth sign has also been ascribed to Thomas Sydenham[9] and Virchow.[4][5]
Redness and heat are due to increased blood flow at body core temperature to the inflamed site; swelling is caused by accumulation of fluid; pain is due to release of chemicals that stimulate nerve endings. Loss of function has multiple causes.[5]
These five signs appear when acute inflammation occurs on the body's surface, whereas acute inflammation of internal organs may not result in the full set. Pain only happens where the appropriate sensory nerve endings exist in the inflamed area—e.g., acute inflammation of the lung (pneumonia) does not cause pain unless the inflammation involves the parietal pleura, which does have pain-sensitive nerve endings.[5]
Process of acute inflammation

The process of acute inflammation is initiated by cells already present in all tissues, mainly resident macrophages, dendritic cells, histiocytes, Kupffer cells and mastocytes. At the onset of an infection, burn, or other injuries, these cells undergo activation and release inflammatory mediators responsible for the clinical signs of inflammation. Vasodilation and its resulting increased blood flow causes the redness (rubor) and increased heat (calor). Increased permeability of the blood vessels results in an exudation (leakage) of plasma proteins and fluid into the tissue (edema), which manifests itself as swelling (tumor). Some of the released mediators such as bradykinin increase the sensitivity to pain (hyperalgesia, dolor). The mediator molecules also alter the blood vessels to permit the migration of leukocytes, mainly neutrophils, outside of the blood vessels (extravasation) into the tissue. The neutrophils migrate along a chemotactic gradient created by the local cells to reach the site of injury.[4] The loss of function (functio laesa) is probably the result of a neurological reflex in response to pain.
In addition to cell-derived mediators, several acellular biochemical cascade systems consisting of preformed plasma proteins act in parallel to initiate and propagate the inflammatory response. These include the complement system activated by bacteria, and the coagulation and fibrinolysis systems activated by necrosis, e.g. a burn or a trauma.[4]
The acute inflammatory response requires constant stimulation to be sustained. Inflammatory mediators have short half lives and are quickly degraded in the tissue. Hence, acute inflammation ceases once the stimulus has been removed.[4]
Exudative component
The exudative component involves the movement of plasma fluid, containing important proteins such as fibrin and immunoglobulins (antibodies), into inflamed tissue. This movement is achieved via the chemically induced dilation and increased permeability of blood vessels, which results in a net loss of blood plasma. The increased collection of fluid into the tissue causes it to swell (edema). This extravasated fluid is funneled by lymphatics to the regional lymph nodes, flushing bacteria along to start the recognition and attack phase of the adaptive immune system system.
Vascular changes
Acute inflammation is characterised by marked vascular changes, including vasodilation, increased permeability and the slowing of blood flow, which are induced by the actions of various inflammatory mediators. Vasodilation occurs first at the arteriole level, progressing to the capillary level, and brings about a net increase in the amount of blood present, causing the redness and heat of inflammation. Increased permeability of the vessels results in the movement of plasma into the tissues, with resultant stasis due to the increase in the concentration of the cells within blood - a condition characterized by enlarged vessels packed with cells. Stasis allows leukocytes to marginate (move) along the endothelium, a process critical to their recruitment into the tissues. Normal flowing blood prevents this, as the shearing force along the periphery of the vessels moves cells in the blood into the middle of the vessel.
Plasma cascade systems
- The complement system, when activated, results in the increased removal of pathogens via opsonisation and phagocytosis.
- The kinin system generates proteins capable of sustaining vasodilation and other physical inflammatory effects.
- The coagulation system or clotting cascade which forms a protective protein mesh over sites of injury.
- The fibrinolysis system, which acts in opposition to the coagulation system, to counterbalance clotting and generate several other inflammatory mediators.
Plasma derived mediators
* non-exhaustive list
| Name | Produced by | Description |
|---|---|---|
| Bradykinin | Kinin system | A vasoactive protein which is able to induce vasodilation, increase vascular permeability, cause smooth muscle contraction, and induce pain. |
| C3 | Complement system | Cleaves to produce C3a and C3b. C3a stimulates histamine release by mast cells, thereby producing vasodilation. C3b is able to bind to bacterial cell walls and act as an opsonin, which marks the invader as a target for phagocytosis. |
| C5a | Complement system | Stimulates histamine release by mast cells, thereby producing vasodilation. It is also able to act as a chemoattractant to direct cells via chemotaxis to the site of inflammation. |
| Factor XII (Hageman Factor) | Liver | A protein which circulates inactively, until activated by collagen, platelets, or exposed basement membranes via conformational change. When activated, it in turn is able to activate three plasma systems involved in inflammation: the kinin system, fibrinolysis system, and coagulation system. |
| Membrane attack complex | Complement system | A complex of the complement proteins C5b, C6, C7, C8, and multiple units of C9. The combination and activation of this range of complement proteins forms the membrane attack complex, which is able to insert into bacterial cell walls and causes cell lysis with ensuing death. |
| Plasmin | Fibrinolysis system | Able to break down fibrin clots, cleave complement protein C3, and activate Factor XII. |
| Thrombin | Coagulation system | Cleaves the soluble plasma protein fibrinogen to produce insoluble fibrin, which aggregates to form a blood clot. Thrombin can also bind to cells via the PAR1 receptor to trigger several other inflammatory responses, such as production of chemokines and nitric oxide. |
Cellular component
The cellular component involves leukocytes, which normally reside in blood and must move into the inflamed tissue via extravasation to aid in inflammation. Some act as phagocytes, ingesting bacteria, viruses, and cellular debris. Others release enzymatic granules which damage pathogenic invaders. Leukocytes also release inflammatory mediators which develop and maintain the inflammatory response. Generally speaking, acute inflammation is mediated by granulocytes, while chronic inflammation is mediated by mononuclear cells such as monocytes and lymphocytes.
Leukocyte extravasation

Various leukocytes are critically involved in the initiation and maintenance of inflammation. These cells must be able to get to the site of injury from their usual location in the blood, therefore mechanisms exist to recruit and direct leukocytes to the appropriate place. The process of leukocyte movement from the blood to the tissues through the blood vessels is known as extravasation, and can be divided up into a number of broad steps:
- Leukocyte localisation and recruitment to the endothelium local to the site of inflammation – involving margination and adhesion to the endothelial cells: Recruitment of leukocytes is receptor-mediated. The products of inflammation, such as histamine, promote the immediate expression of P-selectin on endothelial cell surfaces. This receptor binds weakly to carbohydrate ligands on leukocyte surfaces and causes them to "roll" along the endothelial surface as bonds are made and broken. Cytokines from injured cells induce the expression of E-selectin on endothelial cells, which functions similarly to P-selectin. Cytokines also induce the expression of integrin ligands on endothelial cells, which further slow leukocytes down. These weakly bound leukocytes are free to detach if not activated by chemokines produced in injured tissue. Activation increases the affinity of bound integrin receptors for ligands on the endothelial cell surface, firmly binding the leukocytes to the endothelium.
- Migration across the endothelium, known as transmigration, via the process of diapedesis: Chemokine gradients stimulate the adhered leukocytes to move between endothelial cells and pass the basement membrane into the tissues.
- Movement of leukocytes within the tissue via chemotaxis: Leukocytes reaching the tissue interstitium bind to extracellular matrix proteins via expressed integrins and CD44 to prevent their loss from the site. Chemoattractants cause the leukocytes to move along a chemotactic gradient towards the source of inflammation.
Cell derived mediators
* non-exhaustive list
| Name | Type | Source | Description |
|---|---|---|---|
| Lysosome granules | Enzymes | Granulocytes | These cells contain a large variety of enzymes which perform a number of functions. Granules can be classified as either specific or azurophilic depending upon the contents, and are able to break down a number of substances, some of which may be plasma-derived proteins which allow these enzymes to act as inflammatory mediators. |
| Histamine | Vasoactive amine | Mast cells, basophils, platelets | Stored in preformed granules, histamine is released in response to a number of stimuli. It causes arteriole dilation and increased venous permeability. |
| IFN-γ | Cytokine | T-cells, NK cells | Antiviral, immunoregulatory, and anti-tumour properties. This interferon was originally called macrophage-activating factor, and is especially important in the maintenance of chronic inflammation. |
| IL-8 | Chemokine | Primarily macrophages | Activation and chemoattraction of neutrophils, with a weak effect on monocytes and eosinophils. |
| Leukotriene B4 | Eicosanoid | Leukocytes | Able to mediate leukocyte adhesion and activation, allowing them to bind to the endothelium and migrate across it. In neutrophils, it is also a potent chemoattractant, and is able to induce the formation of reactive oxygen species and the release of lysosome enzymes by these cells. |
| Nitric oxide | Soluble gas | Macrophages, endothelial cells, some neurons | Potent vasodilator, relaxes smooth muscle, reduces platelet aggregation, aids in leukocyte recruitment, direct antimicrobial activity in high concentrations. |
| Prostaglandins | Eicosanoid | Mast cells | A group of lipids which can cause vasodilation, fever, and pain. |
| TNF-α and IL-1 | Cytokines | Primarily macrophages | Both affect a wide variety of cells to induce many similar inflammatory reactions: fever, production of cytokines, endothelial gene regulation, chemotaxis, leukocyte adherence, activation of fibroblasts. Responsible for the systemic effects of inflammation, such as loss of appetite and increased heart rate. |
Morphologic patterns
Specific patterns of acute and chronic inflammation are seen during particular situations that arise in the body, such as when inflammation occurs on an epithelial surface, or pyogenic bacteria are involved.
- Granulomatous inflammation: Characterised by the formation of granulomas, they are the result of a limited but diverse number of diseases, which include among others tuberculosis, leprosy, sarcoidosis, and syphilis.
- Fibrinous inflammation: Inflammation resulting in a large increase in vascular permeability allows fibrin to pass through the blood vessels. If an appropriate procoagulative stimulus is present, such as cancer cells,[4] a fibrinous exudate is deposited. This is commonly seen in serous cavities, where the conversion of fibrinous exudate into a scar can occur between serous membranes, limiting their function.
- Purulent inflammation: Inflammation resulting in large amount of pus, which consists of neutrophils, dead cells, and fluid. Infection by pyogenic bacteria such as staphylococci is characteristic of this kind of inflammation. Large, localised collections of pus enclosed by surrounding tissues are called abscesses.
- Serous inflammation: Characterised by the copious effusion of non-viscous serous fluid, commonly produced by mesothelial cells of serous membranes, but may be derived from blood plasma. Skin blisters exemplify this pattern of inflammation.
- Ulcerative inflammation: Inflammation occurring near an epithelium can result in the necrotic loss of tissue from the surface, exposing lower layers. The subsequent excavation in the epithelium is known as an ulcer.
Inflammatory disorders
Abnormalities associated with inflammation comprise a large, officially unrelated group of disorders which underlie a vast variety of human diseases. The immune system is often involved with inflammatory disorders, demonstrated in both allergic reactions and some myopathies, with many immune system disorders resulting in abnormal inflammation. Non-immune diseases with etiological origins in inflammatory processes are thought to include cancer, atherosclerosis, and ischaemic heart disease.[4]
A large variety of proteins are involved in inflammation, and any one of them is open to a genetic mutation which impairs or otherwise dysregulates the normal function and expression of that protein.
Examples of disorders associated with inflammation include:
- Acne vulgaris
- Alzheimer's
- Amyotrophic Lateral Sclerosis (ALS) "Lou Gehrig's Disease"
- Anemia
- Arthritis
- Asthma
- ADHD
- Autoimmune diseases
- Cancer
- Celiac Disease
- Chronic prostatitis
- Crohn's Disease
- Diabetes
- Erectile Dysfunction (ED)
- Fibromyalgia
- Glomerulonephritis
- Heart Disease
- Human Immunodeficiency Virus (HIV)
- Hypersensitivities
- Inflammatory bowel diseases
- Interstitial cystitis
- Liver Disease
- Lupus
- Lyme Disease
- Migraines
- Multiple Sclerosis
- Myelitis
- Narcolepsy
- Neuropathy
- Obesity
- Pelvic inflammatory disease
- Poliomyelitis
- Reperfusion injury
- Restless Legs Syndrome
- Rheumatoid arthritis
- Sarcoidosis
- Scleroderma
- Sjögren's Syndrome
- Sleep Apnea
- Small Intestinal Bacterial Overgrowth
- Stroke
- Tourette's Syndrome
- Transplant rejection
- Vasculitis
Atherosclerosis
Atherosclerosis, formerly considered a bland lipid storage disease, actually involves an ongoing inflammatory response. Recent advances in basic science have established a fundamental role for inflammation in mediating all stages of this disease from initiation through progression and, ultimately, the thrombotic complications of atherosclerosis. These new findings provide important links between risk factors and the mechanisms of atherogenesis. Clinical studies have shown that this emerging biology of inflammation in atherosclerosis applies directly to human patients. Elevation in markers of inflammation predicts outcomes of patients with acute coronary syndromes, independently of myocardial damage. In addition, low-grade chronic inflammation, as indicated by levels of the inflammatory marker C-reactive protein, prospectively defines risk of atherosclerotic complications, thus adding to prognostic information provided by traditional risk factors. Moreover, certain treatments that reduce coronary risk also limit inflammation. In the case of lipid lowering with statins, this anti-inflammatory effect does not appear to correlate with reduction in low-density lipoprotein levels. These new insights into inflammation in atherosclerosis not only increase our understanding of this disease, but also have practical clinical applications in risk stratification and targeting of therapy for this scourge of growing worldwide importance. http://circ.ahajournals.org/cgi/content/full/105/9/1135
Allergies
An allergic reaction, formally known as type 1 hypersensitivity, is the result of an inappropriate immune response triggering inflammation. A common example is hay fever, which is caused by a hypersensitive response by skin mast cells to allergens. Pre-sensitised mast cells respond by degranulating, releasing vasoactive chemicals such as histamine. These chemicals propagate an excessive inflammatory response characterised by blood vessel dilation, production of pro-inflammatory molecules, cytokine release, and recruitment of leukocytes.[4] Severe inflammatory response may mature into a systemic response known as anaphylaxis.
Other hypersensitivity reactions (type 2 and type 3) are mediated by antibody reactions and induce inflammation by attracting leukocytes which damage surrounding tissue.[4]
Myopathies
Inflammatory myopathies are caused by the immune system inappropriately attacking components of muscle, leading to signs of muscle inflammation. They may occur in conjunction with other immune disorders, such as systemic sclerosis, and include dermatomyositis, polymyositis, and inclusion body myositis.[4]
Leukocyte defects
Due to the central role of leukocytes in the development and propagation of inflammation, defects in leukocyte function often result in a decreased capacity for inflammatory defense with subsequent vulnerability to infection.[4] Dysfunctional leukocytes may be unable to correctly bind to blood vessels due to surface receptor mutations, digest bacteria (Chediak-Higashi syndrome), or produce microbicides (chronic granulomatous disease). Additionally, diseases affecting the bone marrow may result in abnormal or few leukocytes.
Pharmacological
Certain drugs or exogenic chemical compounds are known to affect inflammation. Vitamin A deficiency causes an increase in inflammatory responses,[10] and anti-inflammatory drugs work specifically by inhibiting normal inflammatory components. Certain illicit drugs such as cocaine and ecstasy may exert some of their detrimental effects by activating transcription factors intimately involved with inflammation (e.g. NF-κB).[11][12]
Cancer
Inflammation orchestrates the microenvironment around tumours, contributing to proliferation, survival and migration. Cancer cells use selectins, chemokines and their receptors for invasion, migration and metastasis.[13] On the other hand, many cells of the immune system contribute to cancer immunology, suppressing cancer.
Restless Legs Syndrome
A literature review indicated that 30 of 40 causes of secondary RLS are associated with inflammation. [14]
In light of the fact that many of the highly RLS associated conditions are also associated with inflammatory/immune changes suggests the possibility that RLS may be mediated or affected through these mechanisms. Inflammation is known to cause iron deficiency and hypothetically could cause CNS iron deficiency-induced RLS. Alternatively, an immune reaction to gastrointestinal bacteria or other antigens could contribute to some cases of RLS by a direct immunological attack on the central or peripheral nervous system. Patients with gastrointestinal (GI) diseases such as celiac disease and Crohn’s disease have a high prevalence of RLS and are also associated with inflammation, immune disturbances, iron deficiency and peripheral neuropathy. [15] [16]
A recent study showed an increase in the incidence of small intestinal bacterial overgrowth and irritable bowel syndrome in RLS patients. [17]
Both of these gastrointestinal disorders also have immune and inflammatory disorders. Three studies show evidence that antibiotic therapy may be beneficial for RLS patients who have small intestinal bacterial overgrowth. [18] [19] [20]
Resolution of inflammation
The inflammatory response must be actively terminated when no longer needed to prevent unnecessary "bystander" damage to tissues.[4] Failure to do so results in chronic inflammation, and cellular destruction. Resolution of inflammation occurs by different mechanisms in different tissues. Mechanisms which serve to terminate inflammation include:[4][21]
- Short half-life of inflammatory mediators in vivo.
- Production and release of Transforming growth factor (TGF) beta from macrophages[22][23][24]
- Production and release of Interleukin 10 (IL-10)[25]
- Production of anti-inflammatory lipoxins[26]
- Downregulation of pro-inflammatory molecules, such as leukotrienes
- Upregulation of anti-inflammatory molecules such as the Interleukin 1 receptor antagonist or the soluble tumor necrosis factor receptor (TNFR)
- Apoptosis of pro-inflammatory cells[27]
- Desensitization of receptors
- Increased survival of cells in regions of inflammation due to their interaction with the extracellular matrix (ECM)[28][29]
- Downregulation of receptor activity by high concentrations of ligands
- Cleavage of chemokines by matrix metalloproteinases (MMPs) might lead to production of anti-inflammatory factors.[30]
Acute inflammation normally resolves by mechanisms that have remained somewhat elusive. Emerging evidence now suggests that an active, coordinated program of resolution initiates in the first few hours after an inflammatory response begins. After entering tissues, granulocytes promote the switch of arachidonic acid–derived prostaglandins and leukotrienes to lipoxins, which initiate the termination sequence. Neutrophil recruitment thus ceases and programmed death by apoptosis is engaged. These events coincide with the biosynthesis, from omega-3 polyunsaturated fatty acids, of resolvins and protectins, which critically shorten the period of neutrophil infiltration by initiating apoptosis. Consequently, apoptotic neutrophils undergo phagocytosis by macrophages, leading to neutrophil clearance and release of anti-inflammatory and reparative cytokines such as transforming growth factor-β1. The anti-inflammatory program ends with the departure of macrophages through the lymphatics.[31]
— Charles Serhan
Systemic effects
An infectious organism can escape the confines of the immediate tissue via the circulatory system or lymphatic system, where it may spread to other parts of the body. If an organism is not contained by the actions of acute inflammation it may gain access to the lymphatic system via nearby lymph vessels. An infection of the lymph vessels is known as lymphangitis, and infection of a lymph node is known as lymphadenitis. A pathogen can gain access to the bloodstream through lymphatic drainage into the circulatory system.
When inflammation overwhelms the host, systemic inflammatory response syndrome is diagnosed. When it is due to infection, the term sepsis is applied, with the terms bacteremia being applied specifically for bacterial sepsis and viremia specifically to viral sepsis. Vasodilation and organ dysfunction are serious problems associated with widespread infection that may lead to septic shock and death.
Acute-phase proteins
Inflammation also induces high systemic levels of acute-phase proteins. In acute inflammation, these proteins prove beneficial, however in chronic inflammation they can contribute to amyloidosis.[4] These proteins include C-reactive protein, serum amyloid A, and serum amyloid P, vasopressin, which cause a range of systemic effects including[4]:
- Fever
- Increased blood pressure
- Decreased sweating
- Malaise
- Loss of appetite
- Somnolence
Leukocyte numbers
Inflammation often affects the numbers of leukocytes present in the body:
- Leukocytosis is often seen during inflammation induced by infection, where it results in a large increase in the amount of leukocytes in the blood, especially immature cells. Leukocyte numbers usually increase to between 15 000 and 20 000 cells per microliter, but extreme cases can see it approach 100 000 cells per microliter.[4] Bacterial infection usually results in an increase of neutrophils, creating neutrophilia, whereas diseases such as asthma, hay fever, and parasite infestation result in an increase in eosinophils, creating eosinophilia.[4]
- Leukopenia can be induced by certain infections and diseases, including viral infection, Rickettsia infection, some protozoa, tuberculosis, and some cancers.[4]
Systemic inflammation and obesity
With the discovery of interleukins (IL), the concept of systemic inflammation developed. Although the processes involved are identical to tissue inflammation, systemic inflammation is not confined to a particular tissue but involves the endothelium and other organ systems.
Chronic inflammation is widely observed in obesity.[32] The obese commonly have many elevated markers of inflammation, including[33][34]:
- IL-6 (Interleukin-6)[35][36]
- IL-8 (Interleukin-8)
- IL-18 (Interleukin-18)[35][36]
- TNF-α (Tumor necrosis factor-alpha)[35][36]
- CRP (C-reactive protein)[35][36]
- Insulin[35][36]
- Blood glucose[35][36]
- Leptin[35][36]
Low-grade chronic inflammation is characterized by a two- to threefold increase in the systemic concentrations of cytokines such as TNF-α, IL-6, and CRP.[37] There exist significant correlations between waist circumference and levels of systemic inflammatory response.[38] A predominant factor in this correlation is due to the autoimmune response triggered by adiposity, whereby immune cells may mistake fatty deposits for intruders. The body attacks fat similar to bacteria and fungi. When expanded fat cells leak or break open, macrophages mobilize to clean up and embed into the adipose tissue. Then macrophages release inflammatory chemicals, including TNF-α and (IL-6). TNF's primary role is to regulate the immune cells and induce inflammation. White blood cells then assist by releasing more cytokines. This link between adiposity and inflammation has been shown to produce 10-35% of IL-6 in a resting individual, and this production increases with increasing adiposity.[39]
During clinical studies, inflammatory-related molecule levels were reduced and increased levels of anti-inflammatory molecules were seen within four weeks after patients began a very low calorie diet.[40] The association of systemic inflammation with insulin resistance and atherosclerosis is the subject of intense research.[41]
Studies[which?] have shown in obesity that inflammation and macrophage-specific genes are unregulated in white adipose tissue (WAT). There were also signs of dramatic increase in circulating insulin level, adipocyte lipolysis and formation of multinucleate giant cells. The fat-derived protein called angiopoietin-like protein 2 (Angptl2) elevates in fat tissues. Higher than normal Angptl2 level in fat tissues develop inflammation as well as insulin and leptin resistance. Leptin is a hormone protein secreted by stored fat that signals satiety. Leptin resistance plays a role in the process where appetite overrules the message of satiety. Angptl2 then starts an inflammatory cascade causing blood vessels to remodel and attract macrophages. Angptl2 is an adipocyte-derived inflammatory mediator linking obesity to systemic insulin resistance.[citation needed] It is possible that, as an inflammatory marker, leptin responds specifically to adipose-derived inflammatory cytokines.
C-reactive protein (CRP) is generated at a higher level in obese people. It raises when there is inflammation throughout the body. Mild elevation in CRP increase risk of heart attacks, strokes, high blood pressure, muscle weakness and fragility.[citation needed]
Systemic inflammation and overeating
Hyperglycemia induces IL-6 production from endothelial cells and macrophages.[42] Meals high in saturated fat, as well as meals high in calories irrespective of macronutrient composition, have also been associated with increases in inflammatory markers.[43][44] While the inflammatory responses are acute and arise in response to overeating, the response may become chronic if the overeating is chronic.
Outcomes

The outcome in a particular circumstance will be determined by the tissue in which the injury has occurred and the injurious agent that is causing it. Here are the possible outcomes to inflammation:[4]
- Resolution
The complete restoration of the inflamed tissue back to a normal status. Inflammatory measures such as vasodilation, chemical production, and leukocyte infiltration cease, and damaged parenchymal cells regenerate. In situations where limited or short lived inflammation has occurred this is usually the outcome. - Fibrosis
Large amounts of tissue destruction, or damage in tissues unable to regenerate, can not be regenerated completely by the body. Fibrous scarring occurs in these areas of damage, forming a scar composed primarily of collagen. The scar will not contain any specialized structures, such as parenchymal cells, hence functional impairment may occur. - Abscess Formation
A cavity is formed containing pus, an opaque liquid containing dead white blood cells and bacteria with general debris from destroyed cells. - Chronic inflammation
In acute inflammation, if the injurious agent persists then chronic inflammation will ensue. This process, marked by inflammation lasting many days, months or even years, may lead to the formation of a chronic wound. Chronic inflammation is characterised by the dominating presence of macrophages in the injured tissue. These cells are powerful defensive agents of the body, but the toxins they release (including reactive oxygen species) are injurious to the organism's own tissues as well as invading agents. Consequently, chronic inflammation is almost always accompanied by tissue destruction.
Examples
Inflammation is usually indicated by adding the suffix "-itis", as shown below. However, some conditions such as asthma and pneumonia do not follow this convention. More examples are available at list of types of inflammation.
-
Acute appendicitis
-
Acute dermatitis
-
Acute infective meningitis
-
Acute tonsillitis
Exercise and Inflammation
Exercise-induced acute inflammation
Acute inflammation of the muscle cells, as understood in exercise physiology,[45] can result after induced eccentric and concentric muscle training. Participation in eccentric training and conditioning, including resistance training and activities that emphasize eccentric lengthening of the muscle including downhill running on a moderate to high incline can result in considerable soreness within 24 to 48 hours, even though blood lactate levels, previously thought to cause muscle soreness, were much higher with level running. This delayed onset muscle soreness (DOMS) results from structural damage to the contractile filaments and z-disks, which has been noted especially in marathon runners whose muscle fibers revealed remarkable damage to the muscle fibers after both training and marathon competition. The onset and timing of this gradient damage to the muscle parallels the degree of muscle soreness experienced by the runners.
Z-disks are the point of contact for the contractile proteins. They provide structural support for the transmission of force when the muscle fibers are activated to shorten. However, in marathon runners and those who prescribe to the overload principle to enhance their muscles, show moderate Z-disk streaming and major disruption of the thick and thin filaments in parallel groups of sarcomeres as a result of the force of eccentric actions or stretching of the tightened muscle fibers.
This disruption of the muscle fibers triggers white blood cells to increase following the induced muscle soreness, leading to the inflammatory response observation from the induced muscle soreness. Elevations in plasma enzymes, myoglobinemia, and abnormal muscle histology and ultrastructure are concluded to be associated with the inflammatory response in DOMS. High tension in the contractile-elastic system of muscle results in structural damage to the muscle fiber and plasmalemma and its epimysium, perimysium, and/or endomysium. The mysium damage disrupts calcium homeostasis in the injured fiber and fiber bundles, resulting in necrosis that peaks about 48 hours after exercise. The products of the macrophage activity and intracellular contents (such as histamines, kinins, and K+) accumulate outside the cells. These substances then stimulate the free nerve endings in the muscle; a process that appears accentuated by eccentric exercise, in which large forces are distributed over relatively small cross-sectional area of the muscle.
Post-inflammatory muscle growth and repair
There is a known relationship between inflammation and muscle growth.[46] For instance, high doses of anti-inflammatory medicines (e.g., NSAIDs) are able to blunt muscle growth.[47][48]
It has been further theorized that the acute localized inflammatory responses to muscular contraction during exercise, as described above, are a necessary precursor to muscle growth.[49] As a response to muscular contractions, the acute inflammatory response initiates the breakdown and removal of damaged muscle tissue.[50] Muscles can synthesize cytokines in response to contractions,[51][52][53] such that the cytokines Interleukin-1 beta (IL-1β), TNF-α, and IL-6 are expressed in skeletal muscle up to 5 days after exercise.[50]
In particular, the increase in levels of IL-6 can reach up to one hundred times that of resting levels.[53] Depending on volume, intensity, and other training factors, the IL-6 increase associated with training initiates about 4 hours after resistance training and remains elevated for up to 24 hours.[54][55][56]
These acute increases in cytokines, as a response to muscle contractions, help initiate the process of muscle repair and growth by activating satellite cells within the inflamed muscle. Satellite cells are crucial for skeletal muscle adaption to exercise.[57] They contribute to hypertrophy by providing new myonuclei and repair damaged segments of mature myofibers for successful regeneration following injury- or exercise-induced muscle damage;[58][59][60] high-level powerlifters can have up to 100% more satellite cells than untrained controls.[61][62]
A rapid and transient localization of the IL-6 receptor and increased IL-6 expression occurs in satellite cells following contractions.[54] IL-6 has been shown to mediate hypertrophic muscle growth both in vitro and in vivo.[57] Unaccustomed exercise can increase IL-6 by up to sixfold at 5 hours post-exercise and threefold 8 days after exercise.[63] Also telling is the fact that NSAIDs can decrease satellite cell response to exercise[47], thereby reducing exercise-induced protein synthesis.[48]
The increase in cytokines after resistance exercise coincides with the decrease in levels of myostatin, a protein that inhibits muscle differentiation and growth.[56] The cytokine response to resistance exercise and moderate-intensity running occur differently, with the latter causing a more prolonged response, especially at the 12-24 hour mark.[56]
Chronic inflammation and muscle loss
Both chronic and extreme inflammation are associated with disruptions of anabolic signals initiating muscle growth. Chronic inflammation has been implicated as part of the cause of the muscle loss that occurs with aging.[46][64] Increased protein levels of myostatin have been described in patients with diseases characterized by chronic low-grade inflammation.[65] Increased levels of TNF-α can suppress the AKT/mTOR pathway, a crucial pathway for regulating skeletal muscle hypertrophy,[66] thereby increasing muscle catabolism.[67][68][69] Cytokines may antagonize the anabolic effects of Insulin-like growth factor 1 (IGF-1).[70][71] In the case of sepsis, an extreme whole body inflammatory state, the synthesis of both myofibrillar and sarcoplasmic proteins are inhibited, with the inhibition taking place preferentially in fast-twitch muscle fibers.[72][70] Sepsis is also able to prevent leucine from stimulating muscle protein synthesis.[51] In animal models, when inflammation is created, mTOR loses its ability to be stimulated by muscle growth.[73]
Exercise as a treatment for inflammation
Regular physical activity is reported to decrease markers of inflammation[quantify],[74][75][76] although the correlation is imperfect and seems to reveal differing results contingent upon training intensity. For instance, while baseline measurements of circulating inflammatory markers do not seem to differ greatly between healthy trained and untrained adults,[77][78] long-term chronic training may help reduce chronic low-grade inflammation.[79] On the other hand, levels of inflammatory markers (IL-6) remained elevated longer into the recovery period following an acute bout of exercise in patients with inflammatory diseases, relative to the recovery of healthy controls.[79] It may well be that low-intensity training can reduce resting pro-inflammatory markers (CRP, IL-6), while moderate-intensity training has milder and less-established anti-inflammatory benefits.[77][80][81][82] There is a strong relationship between exhaustive exercise and chronic low-grade inflammation.[83] Marathon running may enhance IL-6 levels as much as 100 times over normal and increases total leuckocyte count and neturophil mobilization.[83] As such, individuals pursuing exercise as a means to treat the other factors behind chronic inflammation may wish to balance their exercise protocol with bouts of low-intensity training, while striving to avoid chronic over-exertion.
Signal-to-noise theory
Given that localized acute inflammation is a necessary component for muscle growth, and that chronic low-grade inflammation is associated with a disruption of anabolic signals initiating muscle growth, it has been theorized that a signal-to-noise model may best describe the relationship between inflammation and muscle growth.[84] By keeping the "noise" of chronic inflammation to a minimum, the localized acute inflammatory response signals a stronger anabolic response than could be achieved with higher levels of chronic inflammation.
See also
- Anaphylatoxin
- Anti-inflammatories
- Essential fatty acid interactions
- Healing
- Interleukin
- Lipoxin
- Substance P
References
- ^ Ferrero-Miliani L, Nielsen OH, Andersen PS, Girardin SE (2007). "Chronic inflammation: importance of NOD2 and NALP3 in interleukin-1beta generation". Clin. Exp. Immunol. 147 (2): 061127015327006––. doi:10.1111/j.1365-2249.2006.03261.x. PMC 1810472. PMID 17223962.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Stedman's Medical Dictionary (Twenty-fifth ed.). Williams & Wilkins. 1990.
- ^ Rather, L. J. (1971). "Disturbance of function (functio laesa): the legendary fifth cardinal sign of inflammation, added by Galen to the four cardinal signs of Celsus". Bull N Y Acad Med. 47 (3): 303–322. PMC 1749862.
- ^ a b c d e f g h i j k l m n o p q r s Cotran (1998). Robbins Pathologic Basis of Disease. Philadelphia: W.B Saunders Company. ISBN 0-7216-7335-X.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b c d Parakrama Chandrasoma, Clive R. Taylor (ca. 2005). "Part A. General Pathology, Section II. The Host Response to Injury, Chapter 3. The Acute Inflammatory Response, sub-section Cardinal Clinical Signs". Concise Pathology (3rd edition (Computer file) ed.). New York, N.Y.: McGraw-Hill. ISBN 0838514995. OCLC 150148447. Retrieved 2008-11-05.
{{cite book}}: Check date values in:|year=(help); External link in|chapterurl=|chapterurl=ignored (|chapter-url=suggested) (help)CS1 maint: year (link) - ^ A Massage Therapist Guide to Pathology Ruth Werner (2009). A massage Therapist Guide to Pathology (4th ed.). Philadelphia, PA and Baltimore, MD: Wolters Kluwer.
- ^ Vogel, Wolfgang H.; Berke, Andreas (2009). Brief History of Vision and Ocular Medicine. Kugler Publications. p. 97. ISBN 90-6299-220-X.
- ^ Porth, Carol (2007). Essentials of pahtophysiology: concepts of altered health states. Hagerstown, MD: Lippincott Williams & Wilkins. p. 270. ISBN 0-7817-7087-4.
- ^ Dormandy, Thomas (2006). The worst of evils: man's fight against pain. New Haven, Conn: Yale University Press. p. 22. ISBN 0-300-11322-6.
- ^ Wiedermann U; et al. (1996). "Vitamin A deficiency increases inflammatory responses". Scand J Immunol. 44 (6): 578–584. doi:10.1046/j.1365-3083.1996.d01-351.x. PMID 8972739.
{{cite journal}}: Unknown parameter|author-separator=ignored (help) - ^ Hargrave, B. Y.; Tiangco, D. A.; Lattanzio, F. A.; Beebe, S. J. (2003). "Cocaine, not morphine, causes the generation of reactive oxygen species and activation of NF-κB in transiently cotransfected heart cells". Cardiovasc Toxicol. 3 (2): 141–151. doi:10.1385/CT:3:2:141. PMID 14501032.
- ^ Montiel-Duarte, C.; Ansorena, E.; López-Zabalza, M. J.; Cenarruzabeitia, E.; Iraburu, M. J. (2004). "Role of reactive oxygen species, glutathione and NF-κB in apoptosis induced by 3,4-methylenedioxymethamphetamine ("Ecstasy") on hepatic stellate cells". Biochem Pharmacol. 67 (6): 1025–1033. doi:10.1016/j.bcp.2003.10.020. PMID 15006539.
- ^ Coussens, L. M.; Werb, Z. (2002). "Inflammation and cancer". Nature. 420 (6917): 860–867. doi:10.1038/nature01322. PMC 2803035. PMID 12490959.
- ^ Weinstock, LB; Walters, A. (2010). "Restless legs syndrome (RLS) is associated with an increased prevalence of small intestinal bacterial overgrowth: is RLS mediated by inflammatory and immunological mechanisms?". Sleep. 33 (S): A256.
- ^ Weinstock, LB; Walters, A.; Mullen, G.; Duntley, SP (2010). "Celiac disease is associated with restless legs syndrome". Journal of Neurology, Neurosurgery & Psychiatry. 55 (6): 1667–73. PMID 19731029.
- ^ Weinstock, LB; Bosworth, BP; Scherl, EJ; et al. (2010). "Crohn's disease is associated with restless legs syndrome". Inflammatory Bowel Diseases. 16 (2): 275–9. PMID 19575360.
{{cite journal}}: Explicit use of et al. in:|last4=(help) - ^ Walters, A.; Weinstock, LB (2011). "Restless legs syndrome is associated with an increased prevalence of small intestinal bacterial overgrowth and irritable bowel syndrome". Sleep Medicine. 12 (6): 610–3. PMID 21570907.
- ^ Weinstock, LB; Fern, SE; Duntleyl, SP (2008). ""Restless legs syndrome in patients with irritable bowel syndrome: response to small intestinal bacterial overgrowth therapy."". Digestive Diseases and Science. 53 (5): 1252–6. PMID 17934858.
- ^ Weinstock, LB; Walters, A.; Duntley, SP; Zeiss, S.; Lewis, N. (2009). "Antibiotic therapy improves restless legs syndrome: a double blind, controlled study of short course of rifaximin". Sleep Medicine. 10 (S2): S28.
- ^ Weinstock, LB (2010). "Antibiotic therapy may improve idiopathic restless legs syndrome: Prospective, open-label pilot study of rifaximin, a nonsystemic antibiotic". Sleep Medicine. 11 (4): 427. PMID 20042368.
- ^ Eming, S. A.; Krieg, T.; Davidson, J. M. (2007). "Inflammation in wound repair: molecular and cellular mechanisms". Journal of Investigative Dermatology. 127 (3): 514–525. doi:10.1038/sj.jid.5700701. PMID 17299434.
- ^ Ashcroft, G. S.; et al. (1999). "Mice lacking Smad3 show accelerated wound healing and an impaired local inflammatory response". Nat Cell Biol. 1 (5): 260–266. doi:10.1038/12971. PMID 10559937.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ Ashcroft, G. S. (1999). "Bidirectional regulation of macrophage function by TGF-β". Microbes Infect. 1 (15): 1275–1282. doi:10.1016/S1286-4579(99)00257-9. PMID 10611755.
- ^ Werner, F.; et al. (2000). "Transforming growth factor-β1 inhibition of macrophage activation is mediated via Smad3". J Biol Chem. 275 (47): 36653–36658. doi:10.1074/jbc.M004536200. PMID 10973958.
{{cite journal}}: Explicit use of et al. in:|last2=(help)CS1 maint: unflagged free DOI (link) - ^ Sato, Y.; Ohshima, T.; Kondo, T. (1999). "Regulatory role of endogenous interleukin-10 in cutaneous inflammatory response of murine wound healing". Biochem Biophys Res Commun. 265 (1): 194–199. doi:10.1006/bbrc.1999.1455. PMID 10548513.
- ^ Serhan, C. N. (2008). "Controlling the resolution of acute inflammation: a new genus of dual anti-inflammatory and proresolving mediators". J Periodontol. 79 (8 Suppl): 1520–1526. doi:10.1902/jop.2008.080231. PMID 18673006.
- ^ Greenhalgh, D. G. (1998). "The role of apoptosis in wound healing". Int J Biochem Cell Biol. 30 (9): 1019–1030. doi:10.1016/S1357-2725(98)00058-2. PMID 9785465.
- ^ Jiang, D.; et al. (2005). "Regulation of lung injury and repair by Toll-like receptors and hyaluronan". Nat Med. 11 (11): 1173–1179. doi:10.1038/nm1315. PMID 16244651.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ Teder, P.; et al. (2002). "Resolution of lung inflammation by CD44". Science. 296 (5565): 155–158. doi:10.1126/science.1069659. PMID 11935029.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ McQuibban, G. A.; et al. (2000). "Inflammation dampened by gelatinase A cleavage of monocyte chemoattractant protein-3". Science. 289 (5482): 1202–1206. doi:10.1126/science.289.5482.1202. PMID 10947989.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ Serhan CN, Savill J (2005). "Resolution of inflammation: the beginning programs the end". Nat. Immunol. 6 (12): 1191–1197. doi:10.1038/ni1276. PMID 16369558.
- ^ Kershaw, E. E.; Flier, J. S. (2004). "Adipose tissue as an endocrine organ". J Clin Endocrinol Metab. 89 (6): 2548–2556. doi:10.1210/jc.2004-0395. PMID 15181022. Review.
- ^ Bastard J; et al. (2000). "Elevated levels of interleukin 6 are reduced in serum and subcutaneous adipose tissue of obese women after weight loss". J Clin Endocrinol Metab. 85 (9): 3338–3342. doi:10.1210/jc.85.9.3338. PMID 10999830.
{{cite journal}}: Unknown parameter|author-separator=ignored (help) - ^ Mohamed-Ali V; et al. (2001). "beta-Adrenergic regulation of IL-6 release from adipose tissue: in vivo and in vitro studies". J Clin Endocrinol Metab. 86 (12): 5864–5869. doi:10.1210/jc.86.12.5864. PMID 11739453.
{{cite journal}}: Unknown parameter|author-separator=ignored (help) - ^ a b c d e f g Loffreda, S.; et al. (1998). "Leptin regulates proinflammatory immune responses". FASEB J. 12 (1): 57–65. PMID 9438411.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ a b c d e f g Esposito, K.; et al. (2002). "Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress". Circulation. 106 (16): 2067–2072. doi:10.1161/01.CIR.0000034509.14906.AE. PMID 12379575.
{{cite journal}}: Explicit use of et al. in:|last2=(help); zero width space character in|doi=at position 9 (help) - ^ Petersen, A. M.; Pedersen, B. K. (2005). "The anti-inflammatory effect of exercise". J Appl Physiol. 98 (4): 1154–1162. doi:10.1152/japplphysiol.00164.2004. PMID 15772055.
{{cite journal}}: Check|doi=value (help); zero width space character in|doi=at position 4 (help) Review. - ^ Rogowski, O.; et al. (2010). "Waist circumference as the predominant contributor to the micro-inflammatory response in the metabolic syndrome: a cross sectional study". Journal of Inflammation. 26 (7). London: 35. doi:10.1186/1476-9255-7-35. PMC 2919526.
{{cite journal}}: Explicit use of et al. in:|last2=(help)CS1 maint: unflagged free DOI (link) - ^ Mohamed-Ali, V.; et al. (1997). "Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-α, in vivo". Journal of Clinical Endocrinology & Metabolism. 82 (12): 4196–4200. doi:10.1210/jc.82.12.4196. PMID 9398739.
{{cite journal}}: Explicit use of et al. in:|last2=(help) - ^ Clément K; et al. (2004). "Weight loss regulates inflammation-related genes in white adipose tissue of obese subjects". FASEB J. 18 (14): 1657–1669. doi:10.1096/fj.04-2204com. PMID 15522911.
{{cite journal}}: Unknown parameter|author-separator=ignored (help)CS1 maint: unflagged free DOI (link) - ^ M Stitzinger (2007). "Lipids, inflammation and atherosclerosis" (pdf). The digital repository of Leiden University. Retrieved 2 November 2007.
- ^ Shoelson, Steven E., Jongsoon Lee, and Allison B. Goldfine. "Inflammation and Insulin Resistance." Journal of Clinical Investigation 116.7 (2006): 1793-801. Print.
- ^ Blackburn P, Després JP, Lamarche B, Tremblay A, Bergeron J, Lemieux I, Couillard C. Impact of postprandial variation in triglyceridemia on low-density lipoprotein particle size. Metabolism. 2003 Nov;52(11):1379-86.
- ^ van Dijk SJ, Feskens EJ, Bos MB, Hoelen DW, Heijligenberg R, Bromhaar MG, de Groot LC, de Vries JH, Müller M, Afman LA. A saturated fatty acid-rich diet induces an obesity-linked proinflammatory gene expression profile in adipose tissue of subjects at risk of metabolic syndrome. Am J Clin Nutr. 2009 Dec;90(6):1656-64. Epub 2009 Oct 14.
- ^ Wilmore, Jack (2008). Physiology of Sport and Exercise. Champaign, IL: Human Kinetics. pp. 26–36, 98–120, 186–250, 213–218. ISBN 978-0-7360-5583-3.
- ^ a b Toth MJ, Matthews DE, Tracy RP, Previs MJ. Age-related differences in skeletal muscle protein synthesis: relation to markers of immune activation. Am J Physiol Endocrinol Metab. 2005 May;288(5):E883-91. Epub 2004 Dec 21.
- ^ a b Mikkelsen UR, Langberg H, Helmark IC, Skovgaard D, Andersen LL, Kjaer M, Mackey AL. Local NSAID infusion inhibits satellite cell proliferation in human skeletal muscle after eccentric exercise. J Appl Physiol. 2009 Nov;107(5):1600-11. Epub 2009 Aug 27.
- ^ a b Trappe TA, White F, Lambert CP, Cesar D, Hellerstein M, Evans WJ. Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. Am J Physiol Endocrinol Metab. 2002 Mar;282(3):E551-6.
- ^ Marimuthu K, Murton AJ, Greenhaff PL. Mechanisms regulating muscle mass during disuse atrophy and rehabilitation in humans. J Appl Physiol. 2011 Feb;110(2):555-60.
- ^ a b Cannon JG, St Pierre BA. Cytokines in exertion-induced skeletal muscle injury. Mol Cell Biochem. 1998 Feb;179(1-2):159-67. Review.
- ^ a b Lang CH, Hong-Brown L, Frost RA. Cytokine inhibition of JAK-STAT signaling: a new mechanism of growth hormone resistance. Pediatr Nephrol. 2005 Mar;20(3):306-12. Epub 2004 Nov 10. Review.
- ^ Pedersen BK, Toft AD. Effects of exercise on lymphocytes and cytokines. Br J Sports Med. 2000 Aug;34(4):246-51. Review.
- ^ a b Bruunsgaard H, Galbo H, Halkjaer-Kristensen J, Johansen TL, MacLean DA, Pedersen BK. Exercise-induced increase in serum interleukin-6 in humans is related to muscle damage. J Physiol. 1997 Mar 15;499 ( Pt 3):833-41.
- ^ a b McKay BR, De Lisio M, Johnston AP, O’Reilly CE, Phillips SM, Tarnopolsky MA, Parise G. Association of interleukin-6 signalling with the muscle stem cell response following muscle-lengthening contractions in humans. PLoS One. 2009 Jun 24;4(6):e6027.
- ^ MacIntyre DL, Sorichter S, Mair J, Berg A, McKenzie DC. Markers of inflammation and myofibrillar proteins following eccentric exercise in humans. Eur J Appl Physiol. 2001 Mar;84(3):180-6.
- ^ a b c Louis E, Raue U, Yang Y, Jemiolo B, Trappe S. Time course of proteolytic, cytokine, and myostatin gene expression after acute exercise in human skeletal muscle. J Appl Physiol. 2007 Nov;103(5):1744-51. Epub 2007 Sep 6.
- ^ a b Serrano AL, Baeza-Raja B, Perdiguero E, Jardí M, Muñoz-Cánoves P. Interleukin-6 is an essential regulator of satellite cell-mediated skeletal muscle hypertrophy. Cell Metab. 2008 Jan;7(1):33-44.
- ^ Grounds MD, White JD, Rosenthal N, Bogoyevitch MA. The role of stem cells in skeletal and cardiac muscle repair. J Histochem Cytochem. 2002 May;50(5):589-610. Review.
- ^ Hawke TJ, Garry DJ. Myogenic satellite cells: physiology to molecular biology. J Appl Physiol. 2001 Aug;91(2):534-51. Review. Erratum in: J Appl Physiol 2001 Dec;91(6):2414.
- ^ Hawke TJ. Muscle stem cells and exercise training. Exerc Sport Sci Rev. 2005 Apr;33(2):63-8. Review.
- ^ Kadi F, Eriksson A, Holmner S, Butler-Browne GS, Thornell LE. Cellular adaptation of the trapezius muscle in strength-trained athletes. Histochem Cell Biol. 1999 Mar;111(3):189-95.
- ^ Eriksson A, Kadi F, Malm C, Thornell LE. Skeletal muscle morphology in power-lifters with and without anabolic steroids. Histochem Cell Biol. 2005 Aug;124(2):167-75. Epub 2005 Sep 29.
- ^ Mikkelsen UR, Schjerling P, Helmark IC, Reitelseder S, Holm L, Skovgaard D, Langberg H, Kjær M, Heinemeier KM. Local NSAID infusion does not affect protein synthesis and gene expression in human muscle after eccentric exercise. Scand J Med Sci Sports. 2010 Aug 24. [Epub ahead of print]
- ^ Visser M, Pahor M, Taaffe DR, Goodpaster BH, Simonsick EM, Newman AB, Nevitt M, Harris TB. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: the Health ABC Study. J Gerontol A Biol Sci Med Sci. 2002 May;57(5):M326-32.
- ^ Reardon KA, Davis J, Kapsa RM, Choong P, Byrne E. Myostatin, insulin-like growth factor-1, and leukemia inhibitory factor mRNAs are upregulated in chronic human disuse muscle atrophy. Muscle Nerve. 2001 Jul;24(7):893-9.
- ^ Shih, Michael. "Skeletal Muscle Hypertrophy Is Regulated via AKT/mTOR Pathway." BioCarta. Web. 21 Mar. 2011. [1].
- ^ Lang CH, Frost RA. Sepsis-induced suppression of skeletal muscle translation initiation mediated by tumor necrosis factor alpha. Metabolism. 2007 Jan;56(1):49-57.
- ^ Garcia-Martinez C, Lopez-Soriano FJ, and Argiles JM. Acute treatment with tumor-necrosis-factor-alpha induces changes in protein metabolism in rat skeletal-muscle. Molecular and Cellular Biochemistry 125: 11-18, 1993.
- ^ Janssen SPM, Gayan-Ramirez G, Van Den Bergh A, Herijgers P, Maes K, Verbeken E, and Decramer M. Interleukin-6 causes myocardial failure and skeletal muscle atrophy in rats. Circulation 111: 996-1005, 2005.
- ^ a b Lang CH, Frost RA, Vary TC. Regulation of muscle protein synthesis during sepsis and inflammation. Am J Physiol Endocrinol Metab. 2007 Aug;293(2):E453-9. Epub 2007 May 15. Review.
- ^ Jurasinski CV, Vary TC. Insulin-like growth factor I accelerates protein synthesis in skeletal muscle during sepsis. Am J Physiol. 1995 Nov;269(5 Pt 1):E977-81.
- ^ Vary TC, Kimball SR. Regulation of hepatic protein synthesis in chronic inflammation and sepsis. Am J Physiol. 1992 Feb;262(2 Pt 1):C445-52.
- ^ Lang CH, Frost RA, Bronson SK, Lynch CJ, Vary TC. Skeletal muscle protein balance in mTOR heterozygous mice in response to inflammation and leucine. Am J Physiol Endocrinol Metab. 2010 Jun;298(6):E1283-94. Epub 2010 Apr 13.
- ^ Smith JK, Dykes R, Douglas JE, Krishnaswamy G, Berk S. Long-term exercise and atherogenic activity of blood mononuclear cells in persons at risk of developing ischemic heart disease. JAMA. 1999 May 12;281(18):1722-7.
- ^ McFarlin BK, Flynn MG, Phillips MD, Stewart LK, Timmerman KL. Chronic resistance exercise training improves natural killer cell activity in older women. J Gerontol A Biol Sci Med Sci. 2005 Oct;60(10):1315-8.
- ^ Stewart LK, Flynn MG, Campbell WW, Craig BA, Robinson JP, McFarlin BK, Timmerman KL, Coen PM, Felker J, Talbert E. Influence of exercise training and age on CD14+ cell-surface expression of toll-like receptor 2 and 4. Brain Behav Immun. 2005 Sep;19(5):389-97.
- ^ a b Gleeson M. Immune system adaptation in elite athletes. Curr Opin Clin Nutr Metab Care. 2006 Nov;9(6):659-65. Review.
- ^ Pedersen BK, Hoffman-Goetz L. Exercise and the immune system: regulation, integration, and adaptation. Physiol Rev. 2000 Jul;80(3):1055-81. Review.
- ^ a b Ploeger HE, Takken T, de Greef MH, Timmons BW. The effects of acute and chronic exercise on inflammatory markers in children and adults with a chronic inflammatory disease: a systematic review. Exerc Immunol Rev. 2009;15:6-41. Review.
- ^ Nicklas BJ, Hsu FC, Brinkley TJ, Church T, Goodpaster BH, Kritchevsky SB, Pahor M. . Exercise training and plasma C-reactive protein and interleukin-6 in elderly people. J Am Geriatr Soc. 2008 Nov;56(11):2045-52.
- ^ Timmerman KL, Flynn MG, Coen PM, Markofski MM, Pence BD. Exercise training-induced lowering of inflammatory (CD14+CD16+) monocytes: a role in the anti-inflammatory influence of exercise? J Leukoc Biol. 2008 Nov;84(5):1271-8. Epub 2008 Aug 12.
- ^ Mackinnon LT. Chronic exercise training effects on immune function. Med Sci Sports Exerc. 2000 Jul;32(7 Suppl):S369-76. Review.
- ^ a b Suzuki K, Nakaji S, Yamada M, Liu Q, Kurakake S, Okamura N, Kumae T, Umeda T, Sugawara K. Impact of a competitive marathon race on systemic cytokine and neutrophil responses. Med Sci Sports Exerc. 2003 Feb;35(2):348-55.
- ^ Pilon, Brad. "Inflammation Affects Your Ability to Build Muscle." Inflammation Theory | Inflammation, Chronic Inflammation, Muscle Building, Health. Web. 27 Mar. 2011. <http://www.inflammationtheory.com/#2>.
External links
- What You Need to Know About Inflammation at Cleveland Clinic
- Anti-Inflammatory Diet - Foods and Inflammation at About.com
- Inflammation at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Information on Natural Anti Inflammatory - Herbs, Diets and Inflammation